Introduction to Ulysses Directory
ulysses_directory.RmdIntroduction
Running an OHDSI study is exciting effort that can lead to meaningful
new evidence on clinical questions across a network of study
participants who have data mapped to the OMOP CDM. However, there are a
lot of tasks to keep track to make an OHDSI study successful. We
introduce the Ulysses R package, as a tool to assist in the
development and organization of an OHDSI study. The idea of the
Ulysses package is inspired by the usethis package that
is used to assist in the development workflow of novel R packages and
projects alike. Similar to the development of an R package, there are
several steps and pieces of documentation needed in an OHDSI study to
effectively run the study across the OHDSI network. By providing
functions that automate tasks and provide consistent structure to OHDSI
studies, Ulysses attempts to help users develop and
communicate new OHDSI studies.
Directory Structure
The first step towards assisting OHDSI studies is to introduce a
consistent directory structure that contains also necessary components
towards executing a study and is easy to follow. Below is a proposed
directory structure, offered by the Ulysses package for
OHDSI studies.
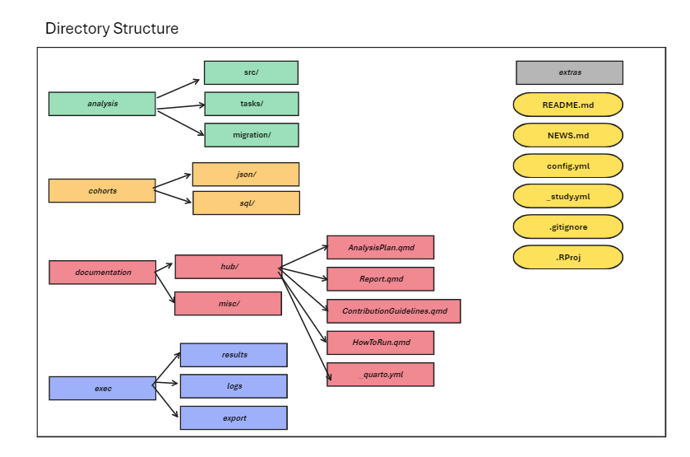
Ulysses Style OHDSI Study Directory
Analysis Folder
The analysis folder contains files that are required for running an
OHDSI study. There are three sub-folders: private,
settings and studyTasks. The studyTasks
folder contains files needed to run the OHDSI study. These could be
several files in a pipeline (i.e. 01_buildCohorts.R,
02_buildStrata.R,
03_baselineCharacteristics.R) or a single strategus json
file that contains details of the modules to run. Next, the
settings folder contains any files that provide details of the
analysis settings. For example, this folder could contain scripts that
specify the settings of an incidence analysis to run in the study.
Likewise, this folder could contain the
createStrategusAnalysisSpecification.R which creates the
strategus json to run the analysis. Finally, the private folder
contains any internal files needed to run the analysis. For example this
could include internal functions to run a study script. The
Ulysses package offers functions to help develop components
of the analysis such as:
-
makeAnalysisScript: initializes an organized .R file, pre-rendered with details about the analysis. -
makeInternals: creates a .R file used for developing internal functions.
Cohorts to Create Folder
OHDSI studies revolve around generated cohort definitions used to
enumerate persons with a particular clinical occurrence (i.e. persons
prescribed ACE Inhibitors for first time). Keeping track of these cohort
definitions, is very important for a successful OHDSI study. Clinical
phenotypes often change during the development of studies, so it is very
important to keep the latest cohort definition json files organized. The
cohortsToCreate folder stores all the json files of cohort
definitions used in the study. They are organized in numbered folders
that are listed at the developers description. By default,
Ulysses creates a starting 01_target folder to
store the target cohort definitions of the study. Ulysses
offers functions that support the organization of this folder, such
as:
-
makeCohortFolder: initializes a new folder to store cohort definitions, i.e. a new folder for comparator cohorts -
makeCohortDetails: a markdown file that provides “plain english” descriptions of the cohort definitions and tracks updates.
Documentation Folder
An OHDSI study consists of lots of documentation that effectively communicate what the study is, how to run it and how to participate. There are three key files stored in this folder:
-
Study Protocol: the protocol offers guidance on the
scientific methods used to conduct the study and justification for why
scientific decisions were made.
Ulyssesauto-generates a skeleton file viamakeOhdisProtocolormakePassProtocol. -
How To Run: a file is needed that provides all
technical specifications and instructions needed to run the study at a
site. This file provides information on how to download the OHDSI study,
prepare the environment for running the study, executing the study and
how to dissiminate the results to the study host.
Ulyssesauto-generates a skeleton file viamakeHowToRun -
Contribution Guidelines: a file is required to
communicate how others can contribute to the study. Contributions can
range from simply running the study to providing significant
development. OHDSI studies require transparency to nodes on what are the
rules and expectations for meaningful contribution to the study.
Ulyssesauto-generates a skeleton file viamakeContribution
There exist scenarios when a full-fledged protocol is not required
for a study. While a study protocol is not required by the institution
running the study, it is still good practice to provide guidance on the
scientific decision making for the study. Ulysses offers a
skeleton file called the Study SAP, implemented via
makeStudySAP, that gives structure to the methods and
rationale for the study while not being as formal as a study
protocol.
The documentation folder may also contain other files that are essential for communicating important aspects of the study.
Results Folder
When an OHDSI study is executed, we require a location to store the
results in an organized fashion. These results can be easily zipped and
sent to the study host. Ulysses initializes a
results folder that can be used as a target for the output. The
results folder is automatically added to the .gitignore so
that results are not accidentally committed to github repository of the
OHDSI study. We intend to add functions to compliment the results folder
in the future.
Logs Folder
Running an OHDSI study is like executing a pipeline of tasks. It is
vital that we know what is going on in the pipeline, whether an error
has occurred or when an execution has taken place. Loggers are an
important part of a pipeline and likewise an OHDSI study.
Ulysses offers a folder to save logs in a single location.
The log folder is is automatically added to the .gitignore
so that results are not accidentally committed to github repository of
the OHDSI study.
Extras Folder
OHDSI studies sometimes contain files that are important to a study
but do not have a natural save location; the extras folder hosts these
files. A prime use for the extras folder is for scripts or files that
are ancillary to the main study. For example scripts such as
KeyringSetup.R are helpful for running the study but not
core to the study itself. Ulysses offers functions that
support the extras folder.
_study.yml
The final file initiated by Ulysses is the
_study.yml file. This is a meta file that provides an
overview of the study. It contains information about who is the study
lead, the date the study started and the full name of the study. We plan
to expand upon this meta file as we feel quality meta data for a study
is useful for 1) automating start-up tasks and 2) providing records to
users.